Why standardisation matters
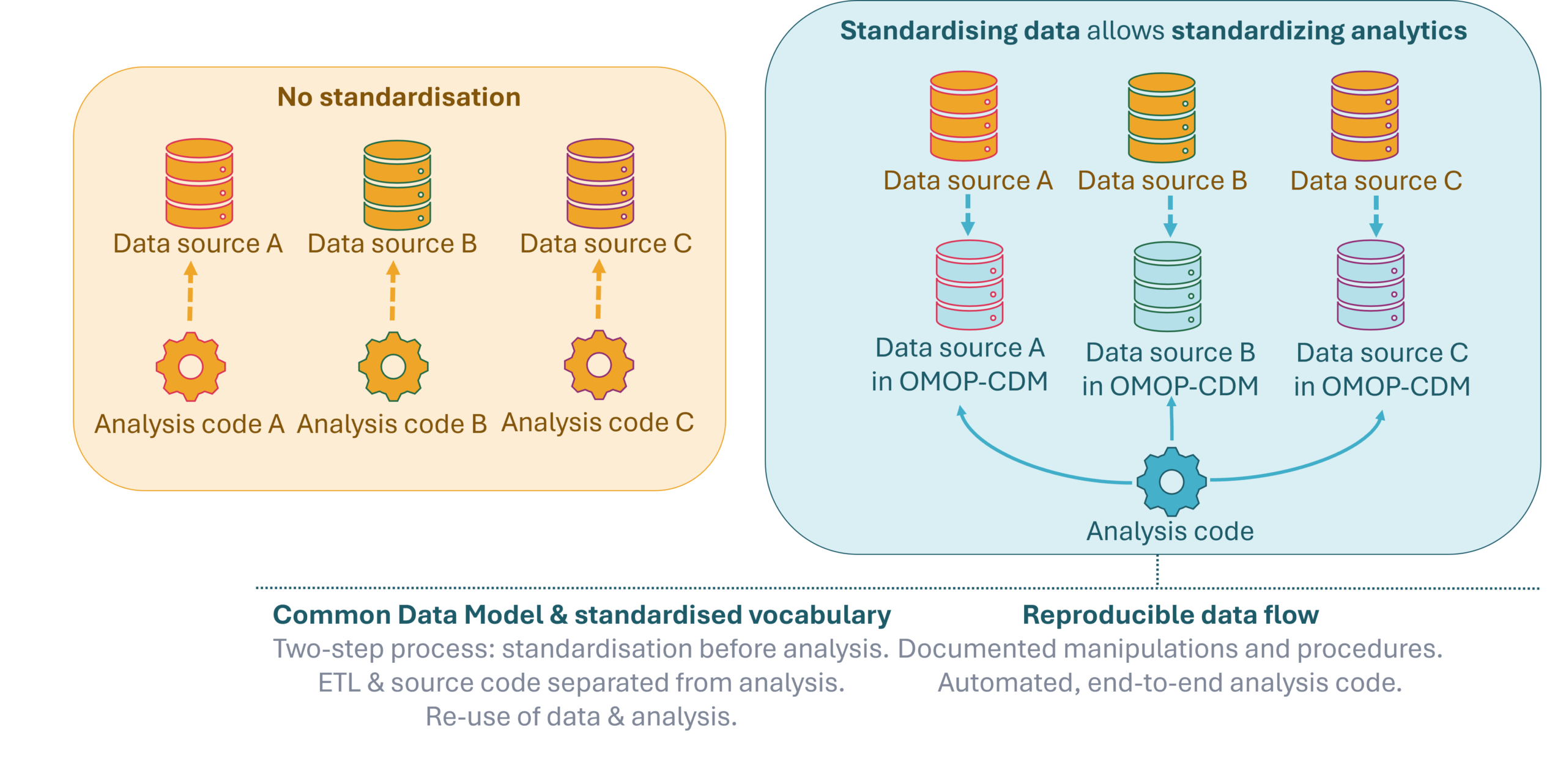
Reliable real‑world evidence depends on more than access to data. Health data across Europe are fragmented, heterogeneous, and stored in different formats, making analyses difficult to reproduce, compare, and scale. The EHDEN Foundation, before an IMI project, addresses this challenge by building on standardised data structures and standardised analytics, enabling robust, transparent, and reusable evidence generation across multiple data sources and countries.
By bringing data to the same structure and applying shared analytical methods, studies can be replicated across settings, results can be compared consistently, and evidence can be generated efficiently while keeping data locally controlled.
The OMOP Common Data Model: a shared language for health data
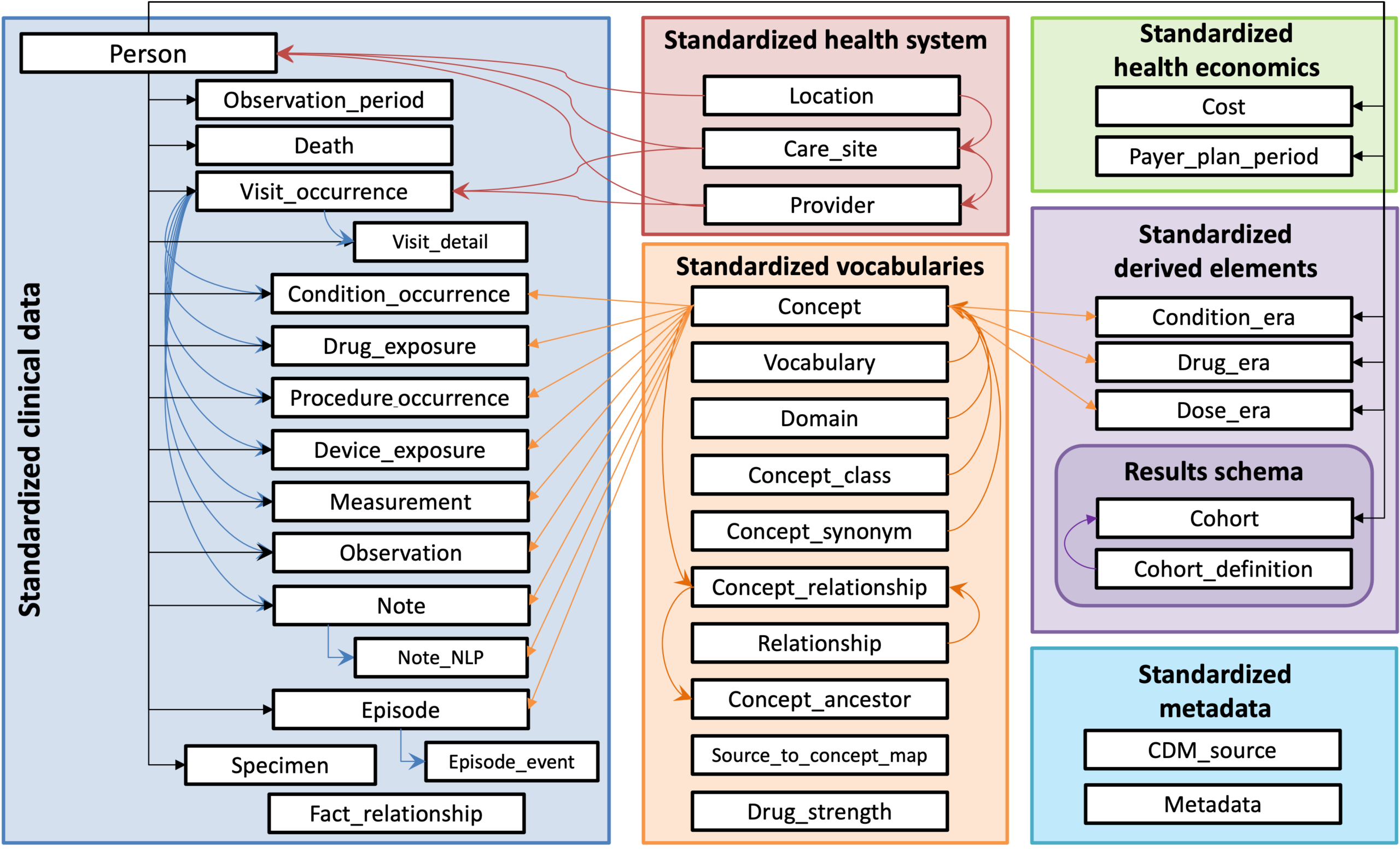
At the core of EHDEN’s approach is the OMOP Common Data Model (OMOP‑CDM), an open, community‑developed data standard created within the global OHDSI (Observational Health Data Sciences and Informatics) collaboration. The OMOP‑CDM provides a common structure, vocabulary system, and analytical conventions that allow diverse healthcare data — such as electronic health records, registries, and claims data — to be analysed in a consistent way.
OMOP does not centralise data. Instead, data remain with the original data holders, mapped locally to the same model. This federated approach respects data protection and governance requirements while making large‑scale analyses possible across institutions and countries. By using standardised vocabularies and a shared structure, OMOP enables reusable study definitions, reusable analytical code, reproducible results, and meaningful comparison across data sources.
Creating value through standardised analytics
Standardised data unlocks value only when paired with standardised analytics. The EHDEN Foundation builds on a rich ecosystem of open‑source analytical tools and validated methodologies developed within the EHDEN project and collaboratively by the OHDSI community and European initiatives such as DARWIN EU®.
These analytics implement common definitions, methods, and quality checks that can be used for a large variety of studies. This approach reduces duplication of effort, increases efficiency, and supports methodological transparency and scientific validity. Standardised analytics allow studies to be executed consistently across multiple data partners, enabling rapid evidence generation.
Key benefits of using standardised data and analytics
- Comparable analyses across countries and care settings
- Faster study execution through reusable methods
- Transparent, reproducible evidence generation
- Data remain under local stewardship
- Alignment with European and global initiatives